13 14 15 16 17 18 19 20 21 22 23 2425 26 27 28 29 30 31 32 40% of a racem..
5 (669) In stock

Solution For 13 14 15 16 17 18 19 20 21 22 23 2425 26 27 28 29 30 31 32 40% of a racemic mixture of 0.2 moles of N2 and 0.6 moles of H2 react to give NH3 according to the equation, N2( g)+H2( g)
13 14 15 16 17 18 19 20 21 22 23 2425 26 27 28 29 30 31 32 40% of a racemic mixture of 0.2 moles of N2 and 0.6 moles of H2 react to give NH3 according to the equation, N2( g)+H2( g) ₹ pressure. Then the ratio of the final volume to the initial volume of gases is:
Video solution 1: 13 14 15 16 17 18 19 20 21 22 23 2425 26 27 28 29 30 31 32 40% of a racemic mixture of 0.2 moles of N2 and 0.6 moles of H2 react to give NH3 according to the equation, N2( g)+H2( g) ₹ pressure. Then the ratio of the final volume to the initial volume of gases is

2020 integrated report of CEMEX, S.A.B. de C.V.

1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 24

Comorbidity, misdiagnoses, and the diagnostic odyssey in patients

exh991-1q21investormeeti
PPT - 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23

Navy Blue Men's polo shirt (012592)

Counting ppt download
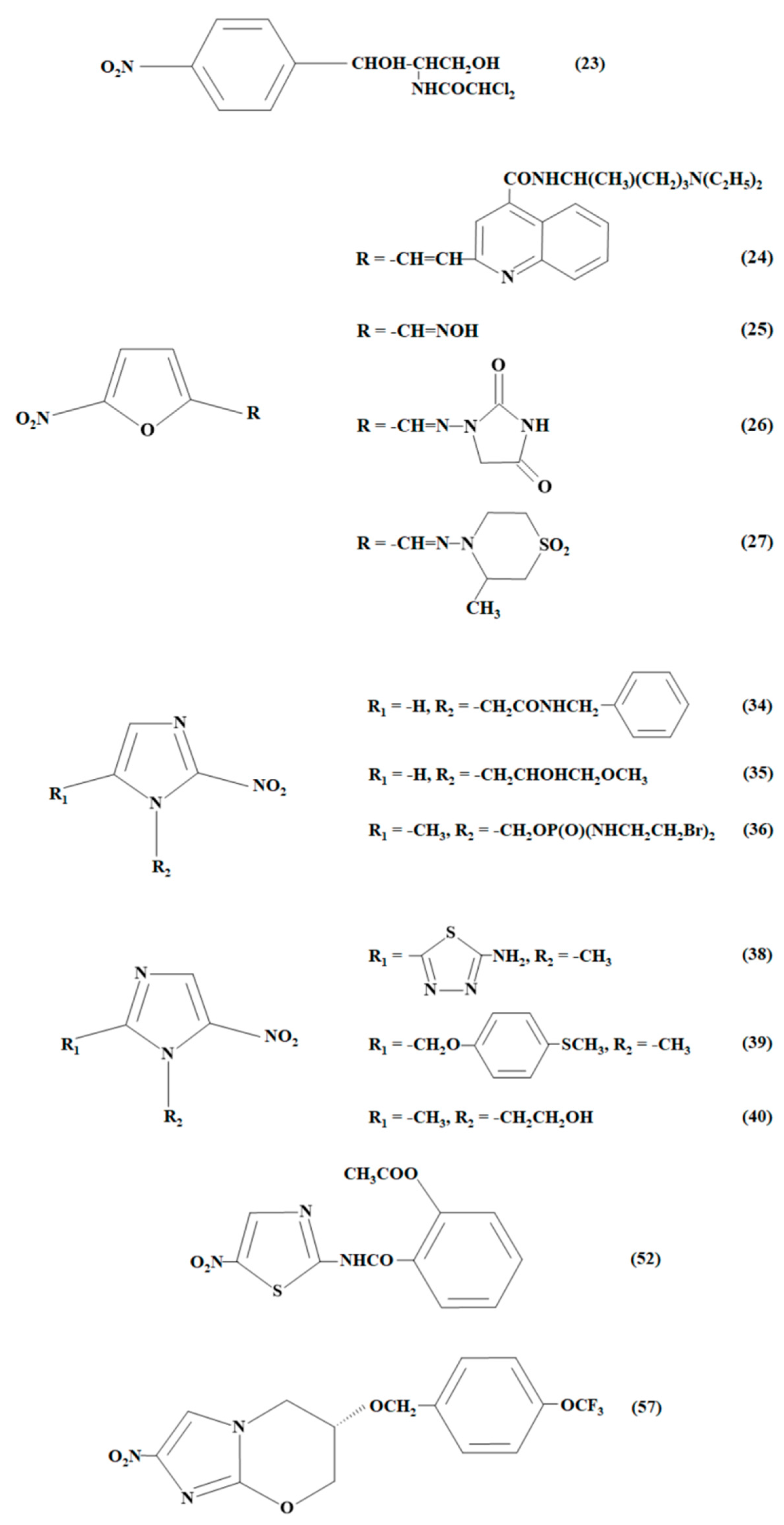
IJMS, Free Full-Text

a et b) Participation en fonction des catégories d'âge au

1 567 16 17 21 22 23 24 25 26 27 28 29 30 31 32 33 34 35 36 37 38
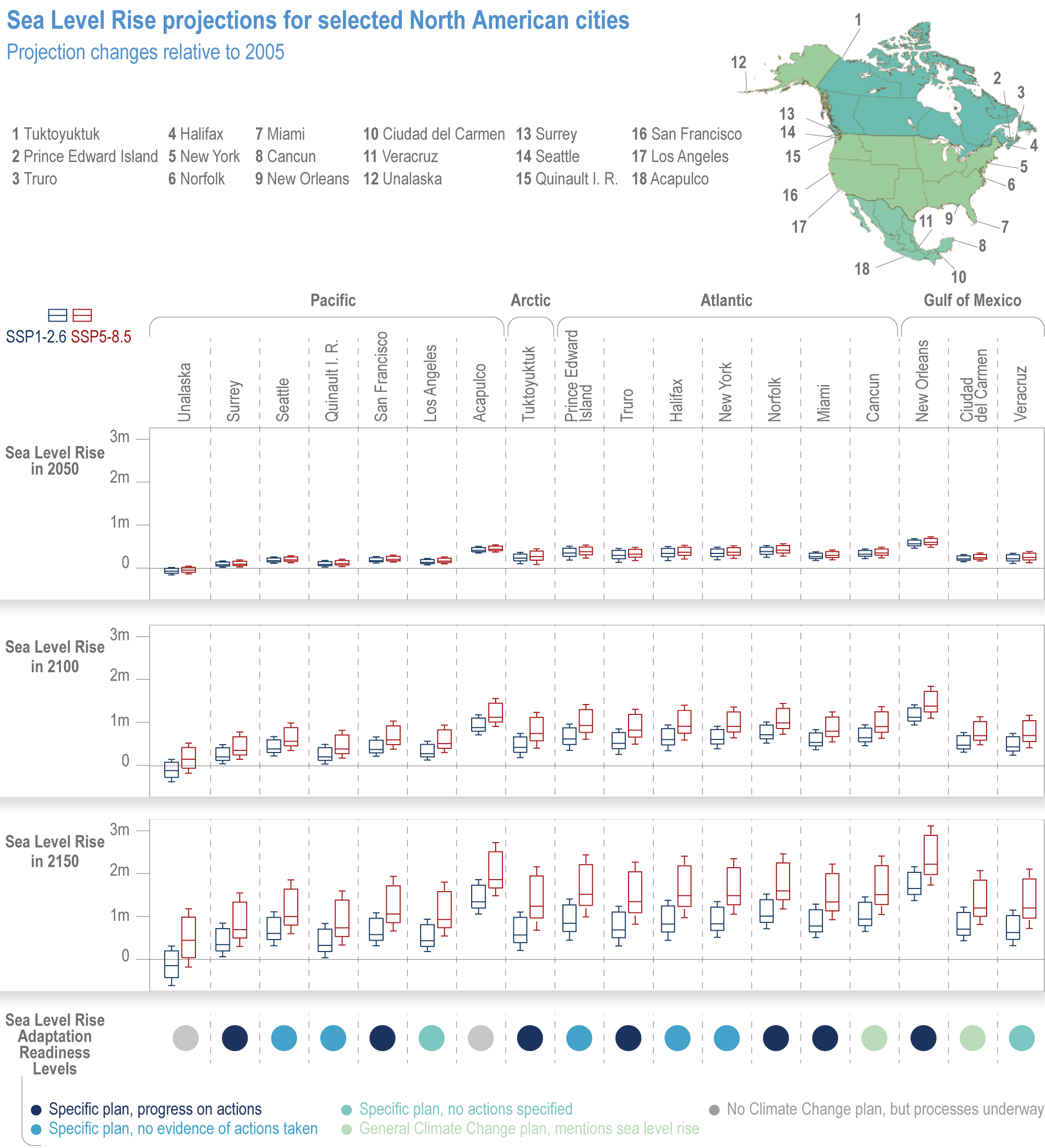
Chapter 14: North America Climate Change 2022: Impacts

Solved J3 Chart Chunks worksheet 1 6 1 9 8 10 11 12 13 14 15
Corsair VENGEANCE 96GB (2x48GB) DDR5 Desktop Memory Kit,, 40% OFF
Word problem involving percentage #viralvideo #viralshorts, #subscribe #viral #r #like
An alloy contains 32% copper, 40% nickel and rest zinc. Find the mass of the zinc is





